COVID-19 PANDEMIC-THE VACCINE-MODERNA: Science: The first batches of Moderna’s coronavirus vaccine being packaged and shipped across the US
Photos show the first batches of Moderna’s coronavirus vaccine being packaged and shipped across the US

- The Food and Drug Administration on Friday granted an emergency use authorization for the COVID-19 vaccine developed by the biotech firm Moderna and the US National Institutes of Health.
- The two-shot Moderna/NIH vaccine was approximately 94.5% effective at preventing COVID-19 in a large clinical trial.
- The FDA last week issued the same authorization for the vaccine created by Pfizer and BioNTech, which had a similar rate of success during trials but is harder to store and transport than the Moderna vaccine.
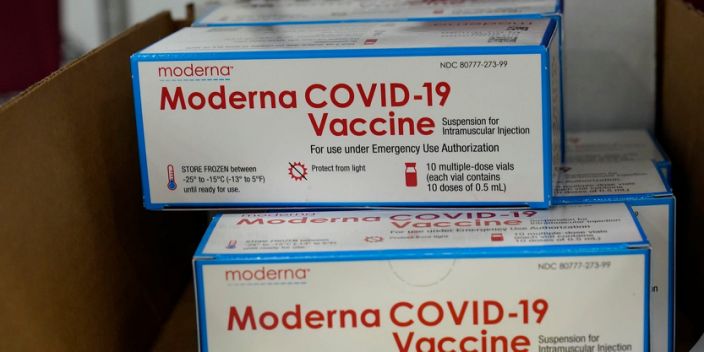
- Photos Sunday showed the vaccine being packed for shipment in Moderna’s distribution facility in Olive Branch, Mississippi, headed for distribution across the US.
- Visit Business Insider’s homepage for more stories.
.

.
The US Food and Drug Administration on Friday issued an emergency use authorization for Moderna’s COVID-19 vaccine, just about a week after it gave the same authorization for a vaccine developed by Pfizer and BioNTech.
The Moderna vaccine was designed in just two days in January 2020 using messenger RNA (mRNA) technology. In clinical trials, the vaccine was found to be approximately 94.5% effective in protecting people against COVID-19, the disease caused by the novel coronavirus.
The vaccine comes at a time when the US is dealing with a surge in COVID-19 cases. The disease has led to more than 316,000 deaths so far in the US this year, according to data from Johns Hopkins University. Globally, it’s killed more than 1.6 million people.
While the vaccine will not bring an end to the ongoing surge, the Centers for Disease Control and Prevention recommends it first be distributed among healthcare workers and among people living in long-term care facilities.
On Sunday, Vivek Murthy, who President-elect Joe Biden intends to nominate as US surgeon general, said the current timeline for coronavirus vaccine distribution was optimistic, adding vaccines may not widely be available until “mid-summer” or “early fall” 2021.


.
SIGN UP TO RECEIVE OUR EMAIL
.
The most important news of the day about the ASEAN Countries and the world in one email: aseanews.net